In 1957, a guy named William Mitchell completely blurred the line between food and science when he created one of the most thought-provoking candies in history—Pop Rocks. You may have tried them, but in case you haven’t, Pop Rocks are little packets of sugar pebbles that pop and fizzle when you put them on your tongue. For some, this can be traumatic.
Have you ever wondered how these explosive little candies work? Just in case, here’s an explanation from me, your science expert (AKA a person who read the Pop Rocks patent).
Pop Rocks are kind of like solid soda, so let’s start with that. Soda is bubbly because carbon dioxide (CO2) gas is forced into the soda’s syrup and water mixture under high pressure.
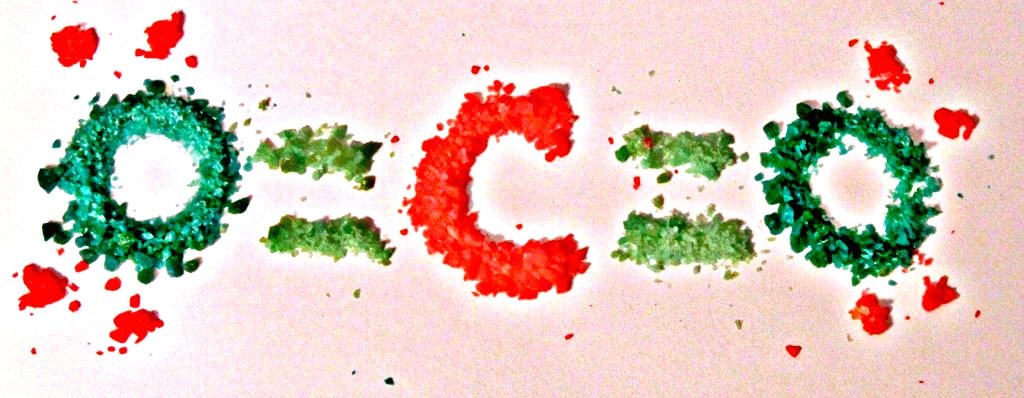
Photo by Sarah Strong
When Pop Rocks are made, high concentrations of CO2 are forced into hot molten sugar under conditions of extremely high pressure. That pressure is maintained as the sugar cools and solidifies. At a certain temperature, the sugar shatters into little “rocks” that still contain a bunch of teeny tiny bubbles of CO2, ready to pop as soon as your saliva dissolves the sugar matrix and sets them free. Cool, right?

photo by Sarah Strong
Pop Rocks were discontinued in the 1980s due to a lack of consumer interest. (Or because of consumer fear. It’s common lore that if you swallow Pop Rocks with soda, your stomach will explode.) According to this article on the history of Pop Rocks, the rights to the scientific recipe behind this carbonated candy traded hands for a couple of decades before they landed back in the hands of its original manufacturers. So now, if you want to experience the food science for yourself, you can find Pop Rocks in retro candy stores like Canditopia, which just opened in downtown New Haven.

